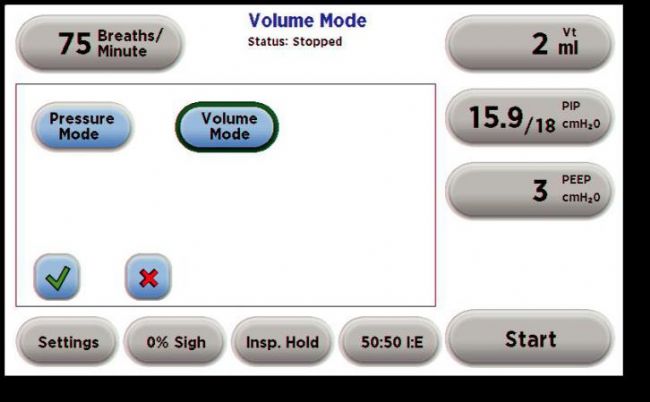
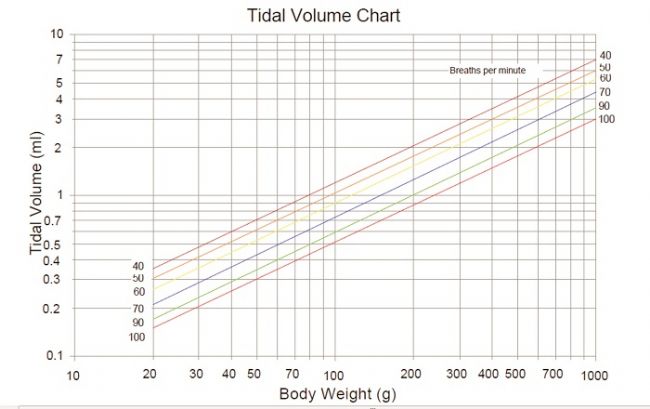
Mechanical ventilation is a method of using a device to restore effective ventilation and improve oxygenation in patients with natural ventilation or oxygenation disorders. The commonly used instruments become "ventilators". Mechanical ventilation is an indispensable means of life support in clinical medical research. It provides time for animal experiments and animal diseases, greatly improving the success rate of some long-term experiments or respiratory failure experiments such as thoracotomy. The history of ventilators is quite old. Here we briefly review: As early as the Roman Empire, the ventilator had a prototype. In 1792, with the support of the Royal Humanne Society, the first tracheotomy, intubation and bellows positive pressure ventilation were used in humans for the recovery of patients with drowning. accept. The latter is terminated because of a fatal pneumothorax. In the 20th century, the "iron lung" developed by Drriker-Shaw in 1928 greatly reduced the mortality rate of polio at that time. A mode of negative pressure ventilation was initiated. Today, we see animal ventilators in different ways of working on the market. So how do they distinguish between them? 1. According to different inhalation control methods, the ventilator can be divided into pressure-controlled ventilator and capacity-controlled ventilator. In the field of clinical ventilator, pressure control is more mainstream. In the field of animal experiments, quite a number of users are accustomed to setting tidal volume for volume control. 2, according to the structural principle of the ventilator, can be divided into piston ventilator and solenoid valve ventilator. The general piston ventilator uses volume control, while the solenoid valve ventilator can perform volume control or pressure control. 3, according to different control methods, can be divided into electric electric control type and pneumatic electric control type ventilator. Because they have different ventilators, they actually give different ways of ventilating the animals. Depending on the ventilation method, we can divide the ventilation methods into: Control mode (CV): The ventilator completely replaces the breathing of the experimental animals. The respiratory rate and tidal volume are controlled by the ventilator and are complete respiratory support. It is suitable for the case where the spontaneous breathing disappears or is very weak. The typical application is the animal's thoracotomy. Control ventilation can be divided into volume control ventilation (VCV) and pressure control ventilation (PCV) depending on the control parameters. Volume Control Ventilation (VCV) is a ventilator that performs ventilation according to preset tidal volume, respiratory rate, and respiratory ratio. Pressure Control Ventilation (PCV) is the ventilator's operation at preset frequencies, breathing ratios, and inspiratory pressures. Auxiliary ventilation (AV): The ventilator's aspiration is triggered by a ventilator detecting a decrease in the airway pressure of the experimental animal. Auxiliary ventilation is a breathing mode similar to clinical application. Animal assisted ventilation can generally be initiated by setting pressure trigger, manual trigger or timed trigger. For animal ventilators, assisted ventilation (AV) is a higher-level breathing mode with only a small number of animal ventilators specific for this function. Sigh mode (sigh): A type of ventilation that takes a deep breath at regular intervals under normal breathing conditions. Usually, in experimental animals, long-term use of controlled ventilation, especially under the condition of volume control ventilation, some marginal alveoli will be inflated, lung function will gradually weaken, adding timing or manual sigh, can prevent lung collapse and improve gas exchange. performance. Let's take a look at how to use it! First, we first look at some physiological parameters of experimental animals, as a reference value for experimental monitoring of animal physiological parameters: Second, tracheal intubation Endotracheal intubation is a common procedure for keeping the animal's respiratory tract smooth during animal surgery and reducing animal suffocation and death. At the same time, endotracheal intubation can also be used for intratracheal drug treatment research. Tracheal intubation is relatively easy to operate on large animals such as dogs and pigs, and relatively difficult for rodents. It is an important step in assisting breathing of animal ventilators. In some experiments requiring ventilator assisted breathing, smooth endotracheal intubation is one of the keys to experimental success. Endotracheal intubation is recommended for use with cannulated indwelling needles: rats typically use a 14-18G cannula, and mice typically use a 20-24G cannula. 1, cut gas tube, direct intubation 2, cut air tube, retrograde guide wire method 3, oral intubation, blind insertion 4, oral intubation, transmitted light assisted method 5, oral intubation, fiber-assisted method Third, select the ventilation mode: select the pressure control mode or the capacity control mode. The use of a pressure-controlled mode is relatively safe because it does not over-inflate the animal's lungs and does not cause damage to the “fragile†lungs; the parameters are easy to set up and operate without regard to the volume and flexibility of the animal's lungs; however, if The experimental time is very long (usually more than 1 hour, it is very likely that accumulation of secretions or other obstructions in the airway of the animal, leading to early termination of inspiratory (the snorkel and airway need to be dissipated). The volume control mode is suitable for long-term experiments because it always delivers a certain volume of gas to the lungs of the animal without causing changes in the flexibility of the lungs of the animal; however, the pre-setting of the parameters is relatively complex and the gas flow rate needs to be determined. Inhalation time (or suction ratio), once these two parameters are set, it is easy to adjust the state of the breathing at any time. Simply raise or lower the gas flow, so that the animal can be finely adjusted. Breathing to maintain a relatively constant CO2 partial pressure. In short, the pressure control mode is fast and safe to operate. It is suitable for short-term animal experiments and ventilator primary users; the volume control mode setting is relatively complicated, but it can finely adjust the animal's breathing, suitable for long-term animal experiments. If the volume control mode is selected, the tidal volume and respiratory rate of the corresponding animal are selected according to the following reference. If the pressure control mode is selected, the initial pumping pressure is generally 15 cmH2O. In order to prevent excessive tidal volume, the animal's lungs are damaged. Generally, the maximum pressure is 20cmH2O. Respiratory non-positive pressure (PEEP): When the end-expiratory airway is open, the airway pressure remains above atmospheric pressure to prevent alveolar atrophy and collapse. Generally, PEEP is required for thoracotomy, and the setting range is 3-5cmH2O. I/E Ratio: The ratio of inspiratory to expiratory time is generally set to 1/2. This parameter cannot be set for some piston ventilators, which is equivalent to 1/1. So that our ventilator setup is complete, we can start experimenting. However, for beginners, the initial parameters may not be clear, we will provide a relevant parameter for your reference. Roller Needle,Cosmetic Roller Needle,Non-Reusable Roller Needle,Disposable Medical Microneedle Changzhou Timerein Biotechnology Co. , Ltd. , https://www.timereinbiotech.com
Capacity control Capacity control and pressure control 

Electric control type Pneumatic electric control type 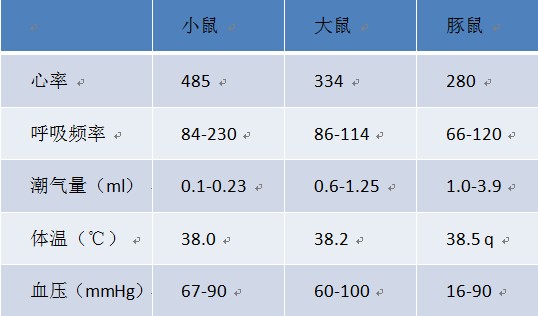
Different laboratories use different intubation techniques, and currently used are: 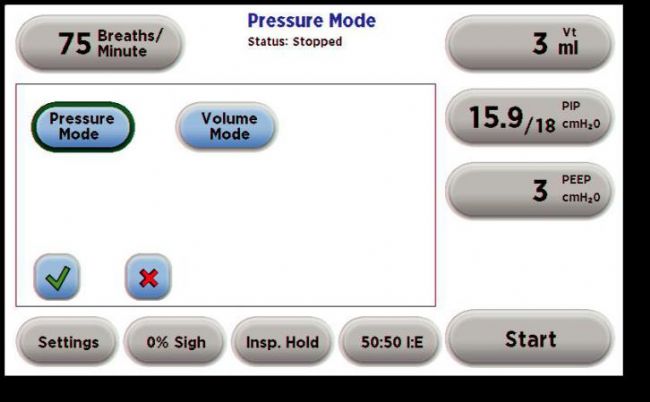
Today, I will talk about the ventilator here first. For more content, please keep an eye on the official website or the public number! ! ! (Search for "Youcheng Bio")