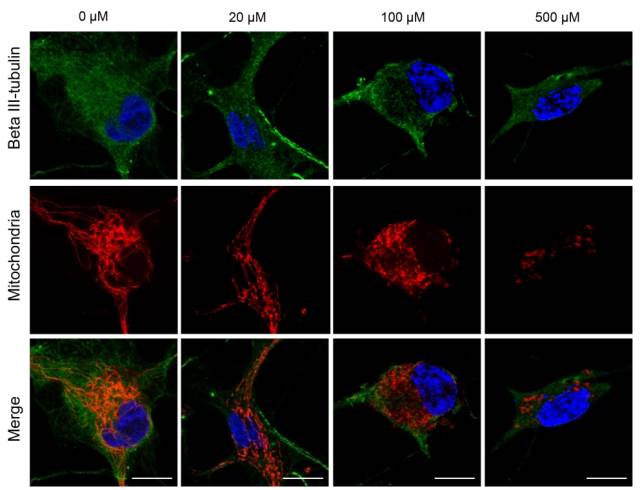
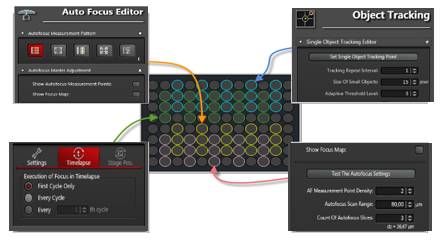
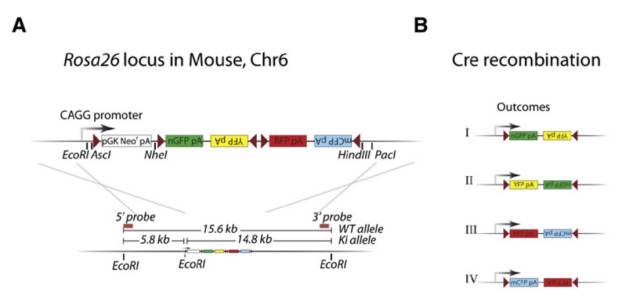
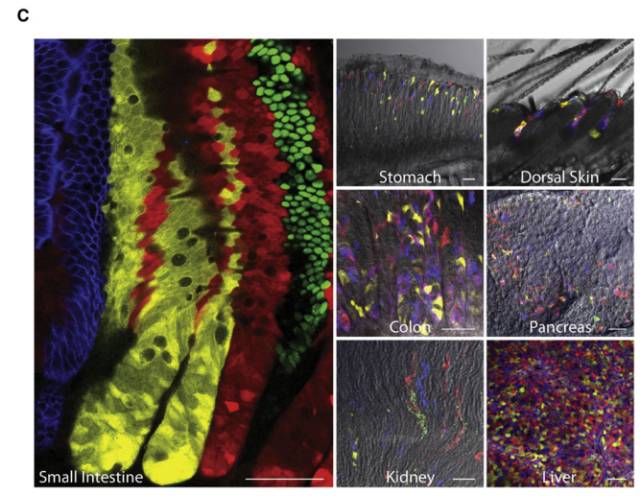
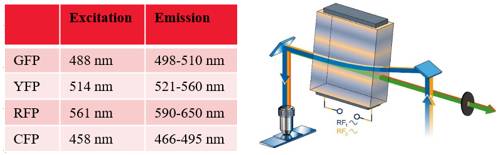
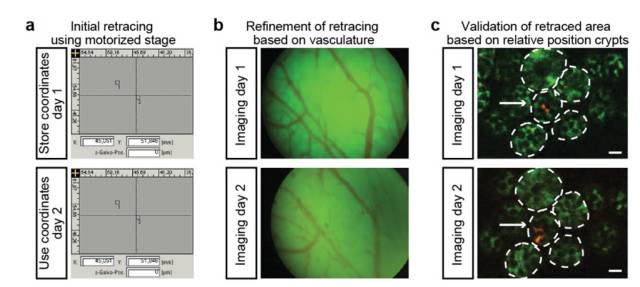
Stem cells are involved in all aspects of individual development, organ transplantation, anti-aging, and cancer treatment. How do individual stem cells divide and differentiate into new cells, tissues or organs? In the adult, how do stem cells fulfill the mission of cell repair and renewal? In the following article, we will describe how to solve these problems in stem cell research by means of microscopic imaging techniques such as confocal and two-photon. Laser confocal scanning microscopy can accurately and controllably perform optical sectioning, clearly presenting sub-structure information of each layer of cells, which is very important for researching protein localization and organelle differentiation. To study the effects of Ketamine on iPSC-induced neuronal mitochondrial morphology, confocal images can be used to present clear images. Figure 1 Effect of Ketamine on iPSC-induced neuronal mitochondrial morphology. (Ketamine Causes Mitochondrial Dysfunction in Human Induced Pluripotent Stem Cell-Derived NeuronsHiroyuki Ito, Tokujiro Uchida, Koshi Makita. PLoS One. 2015; 10(5): 2015 May 28. doi: 10.1371/journal.pone.0128445) This drug stimulation experiment often requires the addition of drugs to living cells and tracking in real time. So how do you keep your cells active? Very simple, just need to reproduce the cell culture environment on the microscope, it can be called Live on the Stage. At present, the Leica LAS X software system provides one-stop service from acquisition and analysis to analysis, and it can also control various environmental conditions such as temperature, humidity and CO2 for cell growth. The following picture shows the common large box culture device and small cell incubator: Figure 2 A commonly used culture device on a microscope. (Left: large box culture device, the whole microscope environment is more stable; right picture: small culture device, easy to operate) Some cell forms and even fate changes take a long time to be reflected, so the stability of the focal plane during the long-term training is particularly important. German quality guarantees the superior stability of the Leica microscope itself, and Leica also offers AFC (Adaptive Focus Control) to track the distance between the bottom of the cell and the objective lens in real time. The focal plane is guaranteed on the hardware. Stable; but what about the focal plane changes caused by cell growth itself? Leica Software Best Focus offers five algorithms for real-time final target cells, whether brightfield or fluorescent. With these hardware and software focal stability, you no longer have to worry about the cells disappearing. Multiple genes or multiple stimulation conditions are often designed in stem cell research, and it is clear that imaging only one sample at a time is not only inefficient but also poor in experimental repeatability. At this point, you can use Leica's multi-point scanning imaging (Mark and Find (for each point imaging requirements are consistent, sample points are not many) or high content imaging (HCS, suitable for multi-point, multi-well plate, responsible for experiments). As shown in the figure below, HCS can not only automatically image multiple holes in a multi-well plate, but also use different imaging conditions for different holes to meet various complex experimental needs. Figure 3 HCS high content imaging. Stem cell imaging studies are required to enter in vivo studies, and multiple methods have been used to track the final differentiation fate of individual stem cells, such as the common R26R-Confetti transgenic strategy. This method involves the expression of four fluorescent proteins, CFP, GFP, YFP and RFP. The excitation and emission spectra of these four fluorescent proteins (Fig. 5, left) are very close, especially GFP and YFP, which are very easy to string. Color, puts stringent requirements on confocal microscopes. Leica adopts patented prism splitting and slit detection system, which can freely adjust the detector's receiving range of fluorescent signals, and collect fluorescent signals with maximum efficiency to avoid cross-color. At the same time, it breaks through the traditional dichroic mirror on the spectroscope. Limited to the filter coating limit, using the completely free AOBS system (Figure 5, right), with the spectral detector, fully realize free detection, to avoid fluorescent cross-color to the greatest extent, directly to obtain experimental results, without the need to use Post-image processing such as software cross-color separation. Figure 4 R26R-Confetti transgene principle and application. Above: R26R-Confetti transgene principle: Brainbow2.1 encoding four fluorescent proteins was inserted into the Rosa26 site. After Cre activation, neomycin was cleaved off. Four fluorescent proteins were randomly expressed in different cells and The localization of the cells is different, GFP nuclear localization, CFP specific expression in the cell membrane, and the other two are cytoplasmic localization. Bottom: Tracking stem cell fate using R26R-Confetti in a variety of tissues. Ruler: 50 um; Pancreas/Kidney/Liver has a ruler of 100 um. (Cell. 2010 Oct 1; 143(1): 134-44. doi: 10.1016/j.cell.2010.09.016. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Snippert HJ1, van der Flier LG, Sato T, van Es JH, van den Born M, Kroon-Veenboer C, Barker N, Klein AM, van Rheenen J, Simons BD, Clevers H.) Figure 5. Excitation and emission spectra of four fluorescent proteins CFP, GFP, YFP and RFP and AOBS system. The microenvironment of stem cell growth is critical to the function and fate of stem cells. Whether it is a 3D environment that simulates stem cell growth in vitro or direct observation of stem cells in vivo, high penetration depth and sensitivity are required for imaging. Multiphoton microscopy Because of its higher penetrability and lower phototoxicity, it has a greater advantage than conventional confocal microscopy. Combined with high-precision stage and software memory function, it can realize long-term tracking of stem cell division and differentiation in vivo. For example, in the experiment of tracking the single cell fate of the small intestine crypt, the experimental mice can not be placed on the microscope stage for continuous culture shooting, then how to track the fate of individual cells? How can I find the original shooting position accurately at the next experimental time? In fact, with the high-precision electric stage and software memory function on the microscope, the parts with significant structure such as the blood vessels of the mouse can be used as reference points, and the original field of view can be found in each photograph (Fig. 6). This can not only reproduce the three-dimensional distribution of stem cells in the entire crypt using a two-photon microscope. It is also possible to connect pictures taken over long days for a long time to restore the fate of a single stem cell. Figure 6 Tracking the field of view of the in vivo imaging. a) The relative coordinates of the imaging field of view can be recorded and stored by the software; b) the vascular structure of the mouse can be used as a reference point; c) the imaging field of view is found and photographed, and the scale is 20 um. (Nature. 2014 Mar 20;507(7492):362-5. doi: 10.1038/nature12972. Epub 2014 Feb 16.) Microscopic imaging technology makes the differentiation and development of stem cells visible in real time. In this paper, ultra-high resolution microscopic imaging technology, which is not mentioned in the space limitation, can clearly reveal the substructure changes in stem cells at the nano level, which is believed to provide another weapon for stem cell research. Dental Amalgamator,Amalgamator Machine,Dental Amalgam Price,Dental Amalgamator Machine ZHEJIANG FOMOS MEDICAL TECHNOLOGY CO.,LTD. , https://www.ifomos.com