Collagen is the most abundant protein in mammals, accounting for 25% to 30% of the total protein. It is widely present in all tissues from the body surface of lower vertebrates to mammalian bodies. Collagen monomers are long cylindrical proteins with a length of about 280 nm and a diameter of 1.4–1.5 nm. It consists of 3 polypeptide chains that are supercoiled around each other. Collagen,Collagen Peptides,Collagen costco,Collagen Peptide Products YT(Xi'an) Biochem Co., Ltd. , https://www.ytlinkherb.com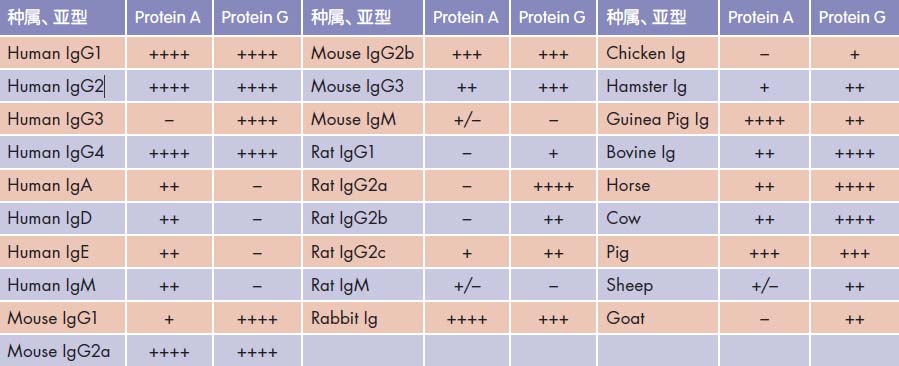
Overview of Protein Affinity Purification
The separation and purification of proteins is an important means to study the structure and function of proteins, and is the only way to prepare industrial enzymes, antibodies, vaccines, genetic recombinant drugs, and the like. There are many ways to purify proteins. Commonly used are the following methods: salting out or isoelectric precipitation method based on different solubility of protein; dialysis and ultrafiltration or gel filtration methods based on protein molecular weight difference; isoelectric focusing based on different designs of proteins Electrophoresis or ion exchange chromatography methods; and affinity chromatography using a specific ligand to bind to a protein of interest, and the like. Compared to nucleic acid purification, the properties of different proteins vary widely, and it is often a difficult and complicated task to explore purification conditions.
Affinity chromatography is a chromatographic method designed by affinity adsorption and dissociation between biomolecules. It has the characteristics of simple operation, mild conditions, and high purity of the obtained protein. It is especially suitable for the purification of active proteins and has low expression. The active protein also has a good separation effect. Commonly used affinity chromatography matrices include: protein A or protein G affinity matrix specific for antibodies, heparin-type affinity matrix for growth factors and nucleic acid binding proteins, purification of tag-containing fusion protein affinity matrix, and binding An affinity matrix of a specific antibody or the like. With the wide application of recombinant gene expression technology, fusion of the target protein with the affinity purification tag and purification of the target protein by affinity chromatography has become the most commonly used technique in the laboratory.
GenStar offers two types of agarose affinity adsorption matrices for the preparation of affinity chromatography columns. One is a metal nickel matrix or a GSH matrix for purifying a fusion protein with a His or GST tag; the other is a protein A and a protein G cross-linked agarose pro for purifying antibodies and immune complexes of various sources. And adsorbing the matrix. Since protein A and protein G have different adsorption capacities for different species or subtypes, please refer to Table 6.1 for the appropriate purification matrix. When it is not possible to discriminate the source or subtype of the antibody, a protein A/G double cross-linking matrix can be used, eliminating the time and material wasted to explore the purification conditions.
Comparison of the relative binding ability of Protein A and Protein G to various antibodies