purpose The enantiomer of 9-hydroxyrisperidone was isolated and this method was applied to monitor the formation of enantiomers in the metabolic process. background Many drug candidates and their metabolites contain one or more chiral centers. Identifying and monitoring the various enantiomers that may be present is a critical step in the drug development process. Supercritical fluid chromatography (SFC) is known to be an efficient technique for chiral separation. In addition, SFC has many advantages, such as high efficiency of analysis, fast separation, and compatibility of solvents with MS. solution In this technical brief, we demonstrate the enantiomeric separation and detection of risperidone, the active metabolite of 9-hydroxyrisperidone, using UPC 2® /MS/MS. The researchers optimized the method using a 9-hydroxyrisperidone standard and applied it to risperidone samples incubated in human liver microsomes. The experiment monitored the conversion process from the parent compound to the hydroxy metabolite. The combination of UPC 2 and MS detection facilitates the enantiomeric separation of 9-hydroxyrisperidone . Figure 1 shows the achiral risperidone and 9-hydroxyrisperidone obtained using a Waters ® Trefoil CEL2 3 × 150 mm, 2.5 μm column with ammonium formate modified methanol as a cosolvent. Optimized chromatogram of the R and S configurations. The method was used to analyze incubation samples quenched at 0, 15, 30, 60, 90 and 120 min to monitor the conversion process from the risperidone precursor to the 9-hydroxy metabolite. Figure 2 shows an example injection in which the two expected peaks of the 9-hydroxy metabolite are located at 3.92 and 4.29 min, respectively. However, at 4.48 and to sum up The combination of UPC 2 and MS detection facilitates the enantiomeric separation of the active metabolite of risperidone, 9-hydroxyrisperidone. Different chiral configurations of isolated compounds are critical steps in drug development. In addition, we have demonstrated that convergent chromatography can be successfully applied to metabolic stability studies. references 1. G Mannens, ML Huang, W Meuldermans, J Hendirckx, R Woestenborghs, J Heykants. Absorption, metabolism, and excretion of risperidone inhumans. Drug Metab Dispos 21: 1134-1141, 1993. 8 Inch Biometric Tablet,Industrial Rugged Android Tablet,Portable Fingerprint Tablet,Rugged Portable Time Attendance Tablet Shenzhen BIO Technology Co.,Ltd. , https://www.hfsecuritytech.com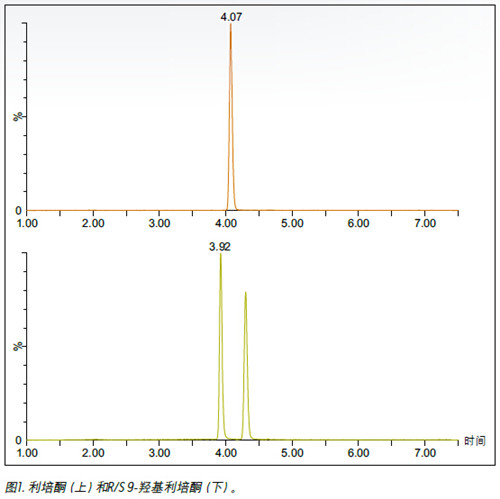
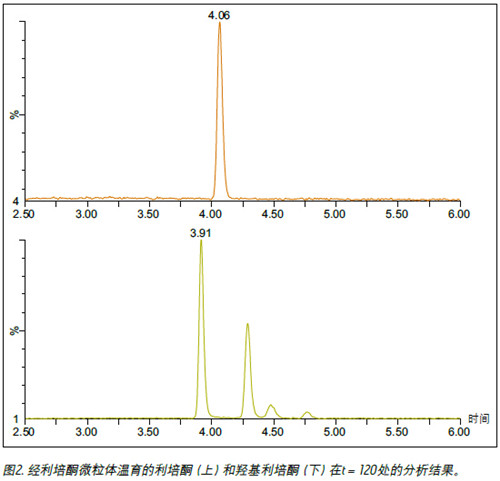
Two additional peaks appeared at 4.76 min, which may be a small amount of 7-hydroxyrisperidone metabolites reported in the literature. 1 We plotted the peak area of ​​the parent compound, risperidone and the four hydroxyl metabolite peaks as a function of time. As shown in Figure 3, most of the parent compound was converted to a metabolite within 30 min. Although the R and S configurations are not absolutely confirmed, according to published data 2 , the process is more prone to form R-9-hydroxyrisperidone. Therefore, we speculate that the earlier eluted peak is in the R configuration and the second peak is in the S configuration. 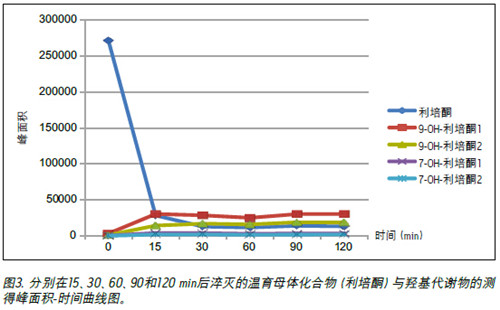
2. N Yasui-Furukori, M Hidestrand, E Spina, G Facciola, MG Scordo, GTybring. Different Enantioselective 9-Hydroxylation of Risperidone by the Two Human CYP2D6 and CYP3A4 Enzymes. Drug Metab Dispos 29:1263-1268, 2001.