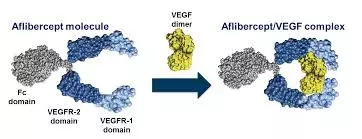
Summary of recent progress in the field of diabetes research (03.21) March 21, 2018 Source: WuXi PharmaTech 1. It is expected to exempt external insulin administration! Beta cell biocarriers have the potential to regulate blood sugar for a long time Recently, researchers from Howard University in Washington, DC, have successfully developed a new type of biomaterial into which beta cells that produce insulin can be implanted. This biomaterial implanted with beta cells reverses diabetes in a mouse model, effectively normalizes glucose levels and significantly increases survival in diabetic mice. The results of the study were recently released at the 100th meeting of the Endocrine Society (ENDO 2018) in Chicago, USA. The beta cells of the pancreas are responsible for the production, storage and release of insulin, a hormone that regulates glucose levels in the blood. During the digestion process, when blood glucose levels begin to rise, beta cells respond quickly, secreting some stored insulin and increasing hormone production. As blood sugar levels drop, so does the amount of insulin secreted by the pancreas. Insulin in diabetics does not regulate blood sugar normally. In patients with type 1 diabetes, beta cells are attacked and destroyed by the autoimmune system; in type 2 diabetes, the response to glucose is impeded and insufficient insulin is produced to control blood glucose levels. ▲Image source 123RF In this study, the researchers developed a novel copolymer microcarrier made of polysaccharide or biomaterial and implanted beta cells from healthy donor mice. The biocarriers implanted with beta cells were transplanted into diabetes. In the mouse model. Tests showed that mice transplanted with biocarriers with beta cells exhibited normalized glucose levels and increased survival compared to untreated groups. The study also found that insulin levels also decreased after normalization of glucose levels after transplantation. The use of biological materials implanted with beta cells does not elicit an immune response. Implanted beta cells promote new blood vessel formation, support long-term survival of transplanted beta cells, and continuous monitoring of blood glucose levels. "Our study proposes a viable and sustainable treatment for diabetes that restores normal blood sugar levels without having to rely on external insulin administration." The first author of the paper, Ms. Diana Elizondo, a graduate student at Howard University, said: "The next step will be Emphasis is placed on the design of donor-compatible beta cells for use in our biomaterials to avoid transplant rejection and to provide long-term insulin recovery therapy." 2. Treatment of diabetic retinopathy, the new drug reached the clinical end point of 3 Regeneron Pharmaceuticals recently announced that the new drug EYLEA (aflibercept) injection for the treatment of moderate to severe non-proliferative diabetic retinopathy (NPDR) reached the primary end point in the phase 3 trial PANORAMA. Diabetic retinopathy is characterized by damage to the retinal microvessels and is a disease usually caused by poor glycemic control in diabetic patients. The disease begins with NPDR and usually has no symptoms or signs. In the United States, approximately 560,000 patients live in moderate to severe NPDR status without diabetic macular edema (DME). As NPDR worsens, blood vessels in the retina become more fragile and leaky, DME may occur, and vision is impaired. NPDR may also progress to the proliferative diabetic retinopathy (PDR) stage, leading to serious, visually cumbersome complications such as vitreous hemorrhage and traction retinal detachment. EYLEA is an ocular injection that is mainly composed of a vascular endothelial growth factor (VEGF) inhibitor. It aims to block the growth of new blood vessels and reduce vascular permeability by blocking two growth factors involved in angiogenesis, VEGF-A and placental growth factor (PLGF). ▲The mechanism of action of EYLEA (Source: EYLEA official website) The FDA-approved drug is supported by research including seven key phase 3 clinical trials. PANORAMA is a continuous, critical, double-blind, randomized, two-year trial involving 402 patients with the goal of investigating the efficacy of EYLEA in patients with severe to severe NPDR without DME compared with sham injection. The primary endpoint was the proportion of patients who experienced a two- or more-score improvement in the Diabetic Retinopathy Severity Scale (DRSS). The first primary endpoint was measured at 24 weeks and the second at 52 weeks. The DRSS assesses the severity of diabetic retinopathy based on retinal photographs after eye dilation. The results showed that 58% of patients with EYLEA had a two- or more improvement in DRSS at 24 weeks, compared with only 6% in the sham-injected group (p < 0.0001). Patients in the treatment group received an average of 4.4 EYELA injections during the first 24 weeks. There are no new security issues in the trial. Patients treated with EYLEA had a mild intraocular inflammation (IOI) (0.085%), consistent with the IOI rate observed in previous clinical trials. "In this first trial of this population, EYLEA demonstrated that it can reverse disease progression in patients with moderate to severe non-proliferative diabetic retinopathy with no diabetic macular edema." Regeneron President and Chief Scientific Officer Dr. George D. Yancopoulos said: "Patients in the trial will continue to evaluate whether EYLEA can prevent neovascular complications or diabetic macular edema that cause vision loss. We look forward to sharing a year later this year. The results of the study." 3. Janssen type 2 diabetes drugs significantly reduce the risk of cardiovascular death Janssen Pharmaceutical Companies of Johnson & Johnson recently announced a new analysis that INVOKANA (canagliflozin) can significantly reduce the risk of cardiovascular death or heart failure hospitalization (HHF) in patients with type 2 diabetes. This exploratory analysis of the CANVAS project was presented at the 67th Annual Scientific Meeting of the American College of Cardiology and published in the journal Circulation. Diabetes is a growing global epidemic. As of 2015, 400 million adults worldwide are affected by diabetes, of which about 100 million are in China. People with type 2 diabetes are resistant to insulin or are unable to secrete enough insulin to maintain normal blood sugar levels. Sustained high glucose levels can lead to complications of diabetes such as heart disease, stroke, kidney failure, neuropathy, lower extremity amputation and blindness. INVOKANA is a daily oral diabetes drug, a new class of drugs that are inhibitors of sodium glucose cotransporter 2 (SGLT2). These drugs reduce the body's blood sugar levels by blocking the kidney's reabsorption of blood sugar and increasing the excretion of blood sugar in the urine. The CANVAS project is the longest and most extensive focus on cardiovascular efficacy in all SGLT2 inhibitors to date, and the first in more than 10,000 patients with a history of cardiovascular disease, or at least two cardiovascular risk factors. Among the patients with type 2 diabetes, the efficacy, safety and persistence of INVOKANA were evaluated. The analysis showed that INVOKANA significantly reduced cardiovascular risk or HHF risk by 22%, reduced heart failure or hospitalization by 30%, and reduced HHF risk by 33% alone. Patients with a history of heart failure had a 39% reduction in cardiovascular death or HHF risk, and patients without a history of heart failure also had a 13% reduction from baseline. The effect of INVOKANA on cardiovascular death or HHF risk can be compared across multiple subgroups. In addition, the data also confirmed that INVOKANA has a proportional effect in patients with type 2 diabetes who have a high risk of cardiovascular disease compared with placebo. Dr. Robert Cuddihy, vice president of medical affairs at Janssen's R&D department, said: "Heart failure and its more serious cardiovascular deaths are key to many doctors and patients managing type 2 diabetes. A new analysis of this landmark CANVAS project will help To strengthen the important clinical benefits of canagliflozin." Reference materials: [1] Beta cell-seeded implant restores insulin production in type 1 diabetes mouse model [2] Regeneron's Eylea Shows Promising Data for Diabetic Retinopathy [3] INVOKANA® (canagliflozin) Significantly Reduces the Risk of Heart Failure-Related Outcomes in Type 2 Diabetes Patients at Risk for or With a History of Cardiovascular Disease Original Title: Summary of Recent Progress in Diabetes Research (No. 54) Disposable Paper Toilet Seat,Disposable Toilet Seat Covers,Disposable Sanitary Toilet Seat Cover,Disposable Paper Seat Cover RFX+CARE Manufacturing Co.,Ltd. , https://www.rk-rfxcare.com