Heyin Fennel,Foeniculum Vulgare Mill,Fennel For Cooking,Organic Fennel Seeds Zhejiang Shanying Trading Co.,Ltd. , https://www.shanyingtrading.com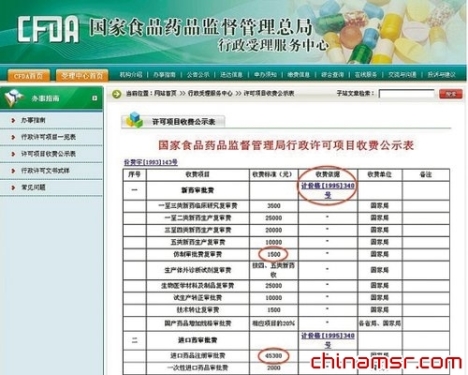 “Similarly registering a generic drug in China, the Chinese local pharmaceutical company only needs RMB 1,500, while the imported drug requires 45,300. The excessively high drug registration fee and the 'unfair' registration condition make high-quality international generic drugs come from Drugs from the United States, Canada, India, etc. are not easy to enter the Chinese market. This result prevents Chinese residents from enjoying cheap and high-quality American and Indian drugs."
“Similarly registering a generic drug in China, the Chinese local pharmaceutical company only needs RMB 1,500, while the imported drug requires 45,300. The excessively high drug registration fee and the 'unfair' registration condition make high-quality international generic drugs come from Drugs from the United States, Canada, India, etc. are not easy to enter the Chinese market. This result prevents Chinese residents from enjoying cheap and high-quality American and Indian drugs."
Recently, there were netizens who claimed to be “American small drug companies†that they thought that excessively high drug registration fees would make it impossible for quality generic and cheap international generic drugs to enter the Chinese market, causing heated discussions. A survey conducted by the Yangcheng Evening News found that the drug registration fee, which was criticized as unfair, has not changed for 18 years.
â— The current status of the drug registration fee has not changed for 18 years. The reporter saw on the State Food and Drug Administration administrative licensing project fee publicity form (see the figure on the right) that the new drug approval is divided into six categories, of which the sixth category is the generic drug that is often referred to. According to the public notice, the approval fee for generic drugs for domestic companies is 1,500 yuan. For imported drugs, regardless of whether they are new drugs or generic drugs, the registration fee for imported drugs is uniformly 45,300 yuan.
However, the reporter also noticed that the above charges were based on the Circular on Adjustment of Drug Approvals and Inspection Fees issued by the State Development Planning Commission and the Ministry of Finance on April 6, 1995 (calculated price [1995] No. 340). The notice has been implemented since May 1, 1995. That is to say, this is still the charging standard of 18 years ago.
What is the concept of 18 years? In 18 years, the National Development and Reform Commission (formerly the State Planning Commission) has initiated more than 30 rounds of drug price adjustments. In 18 years, the average monthly income of Chinese employees rose from about 400 yuan to about 4,000 yuan. It can be said that the 10,000 yuan of that year was almost equivalent to today's 100,000 yuan.
Since this is the case, has the drug registration fee charging standard been lagging behind for 18 years? In the end should not change?
â— Sound changes! Domestic and foreign drug companies must be fairly based on what they claim to be online users of “small US pharmaceutical companiesâ€. The fees and requirements for registration of drugs by foreign drug companies in China are far higher than those of Chinese domestic companies. For example, the same is to register a generic drug in China. The Chinese local drug company only needs 1,500 yuan, while the United States and foreign pharmaceutical companies need 45,300 yuan, and the foreign drug company is 30 times the local drug company. In another example, if the same generic drug is registered, the Chinese local company will apply for the generic generic drug type VI. After the audit, if it meets the requirements, it may directly issue the drug approval number. The imported drugs, regardless of whether they are generic drugs, will be issued to the “Approval Document for Clinical Trials of Drugs†after the audit, and after the completion of the clinical trials, an approval decision will be made.
In response, foreign pharmaceutical companies believe that these violate the provisions of the fair and equitable administrative licensing law. Such fee standards need to be changed and should be paid in accordance with the principles of openness, fairness and fairness.
change! Fees are too low. Another voice also believes that the current charge for the approval of generic drugs is subject to change. The reason is that the fees charged 18 years ago are too low for the time being.
A person from a Beijing-based professional consultancy company engaged in drug registration at home and abroad told the reporter of the Yangcheng Evening News that, according to the "Registration Measures for Drug Registration," the State Administration of Food and Drug Administration can carry out on-site inspections of the development and production of overseas drug companies for approval, and collect samples. . “Although there is no express provision, the actual cost of these on-site inspections is also included in the approval fee. That is to say, the amount of 45,300 yuan paid by foreign pharmaceutical companies contains the official dispatch fee of the approving officers, but the national food The General Drug Administration has not exercised such power."
Industry insiders said that this assignment approval is rarely implemented. It was not until 2011 that drug registration was the first time to conduct on-site inspections of imported pharmaceuticals overseas. At that time, the State Food and Drug Administration formed seven inspection teams and went to seven countries including the United States, France, Italy, India, Hungary, South Korea, and Japan. They used bevacizumab injection, dinoprost injection, and hydrochloric acid for injection. Seven varieties such as gemcitabine carried out overseas on-site inspections and achieved a breakthrough of “zero†in overseas inspections of imported drugs.
“Actually, the cost of overseas on-site production inspections came from public funds, because at the current price level, the 45,300 yuan approval fee received from companies alone is not enough to support offshore production inspections.†The above-mentioned sources frankly stated that "The charge standard 18 years ago is still inappropriate. This is judged by the drug administration department. The industry just thinks that the current charge standard is not high at all."
change! The approval rate is too slow. "The threshold for drug registration is not high. The biggest bottleneck for drug approval is not charging. Instead, the drugs supervision personnel possessed by the drug administration department are seriously inadequate, so that the speed of approval of imported drugs is too slow. Conversely, if the approval is improved, Fees can help bring in talents, speed up the approval process, and increase efficiency. I believe many foreign pharmaceutical companies are willing to raise their prices.†This is a voice from foreign pharmaceutical companies and a third voice.
According to a study by IMS, at the registration stage of pharmaceuticals, the listing of new drugs in China was on average 4-5 years later than other countries because of human resources and process restrictions. Zhang Meng, head of IMS China's regional consulting business, told reporters that in China, the registration approval of innovative drugs will take about one and a half years to obtain a clinical access permit.
Zhuo Yongqing, executive director of the Pharmaceutical Industry Research and Development Industry Committee of the China Association of Enterprises with Foreign Investment, also told reporters that compared with international comparisons, the United States has more than 3,000 NDAs (new drug applications), and the National Food and Drug Administration has only 120 review centers. Individuals, of whom about 80 are auditors. In sharp contrast, according to the Annual Report on Drug Registration Approval issued by the General Administration of Food and Drug Supervision of the State, the total number of drug registration applications received in 2009 was 6,428, and the total number of applications received in 2010 was 6,294, and new drugs were accepted in 2011. 3620 applications for registration.
“This person has to handle more than 6,000 reviews a year and work overtime. These review experts are very painful.†Zhuo Yongqing also called for, “Society in today’s society has five years ago and ten years ago, twenty years ago. With the big changes, the demand for innovative drugs is very large. When the national industrial policy changes, is it right? There should be some changes in the organizational structure of our government."